 Virtual reality surgical simulator
devices designed and built by Roger E. Kaufman.
Virtual reality surgical simulator
devices designed and built by Roger E. Kaufman.
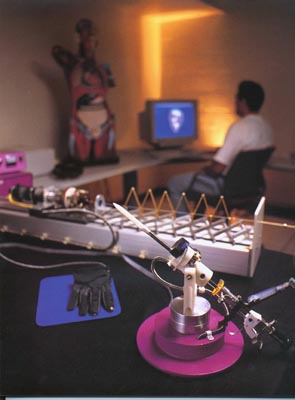
The device with the purple circular base and the stainless
steel surgical instruments is for training urologists. The long
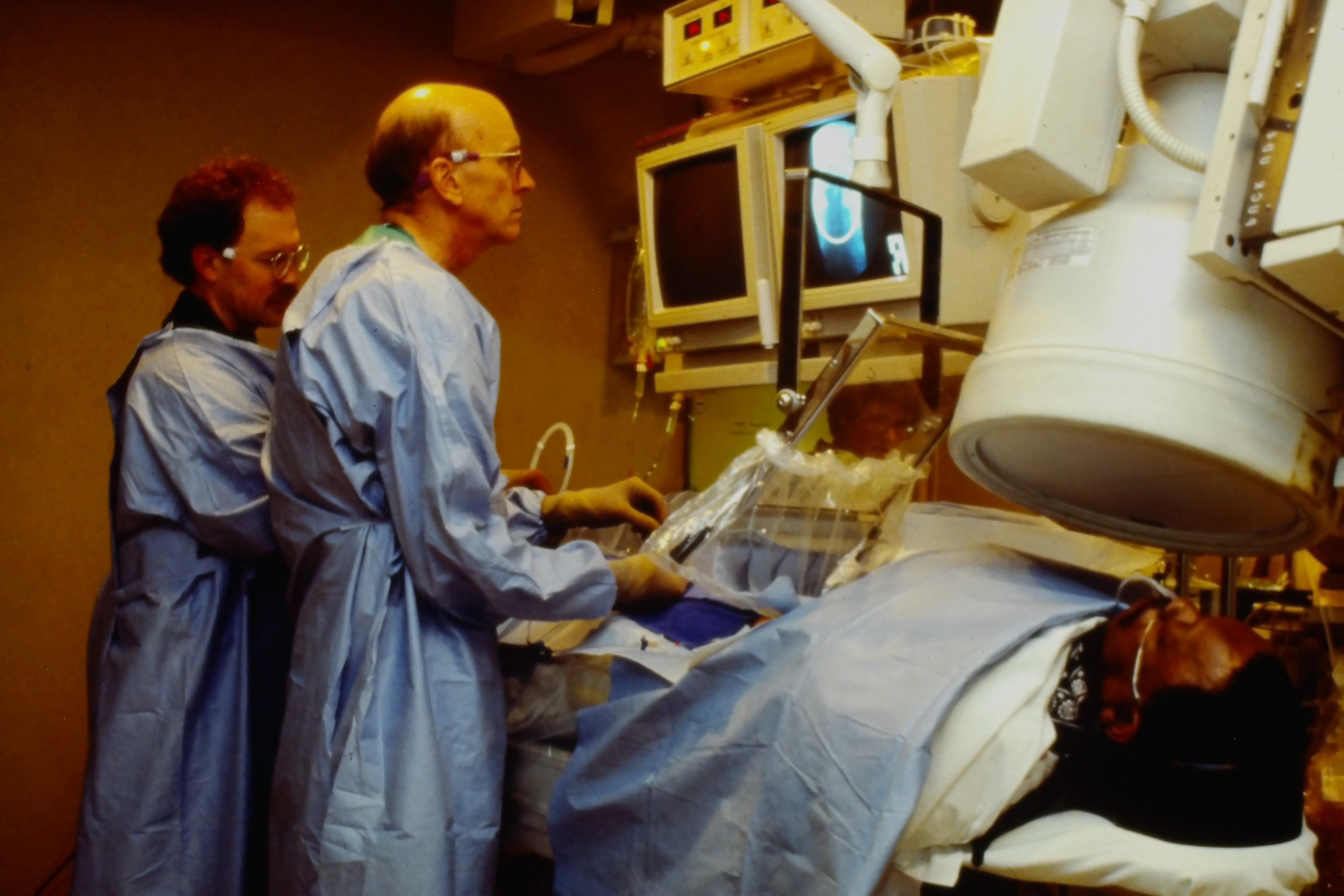
device with the white base is a haptic catheter device for training
interventional radiologists in vena
cava filter placement. An active control system transmits
position and force data to the computer via the interface electronics
in the case on the left and the device replicates the feel and
response of an actual catheter based on the results of the real-time
simulation.
Other devices I designed to train urologists in performing
cryosurgery. With the simulator, urologists could practice a procedure
by varying the entry angle of the cryoprobe in the pitch and yaw
directions and also moving the probe in and out. Simulated tissue
resistance is felt by the user via a computer-controlled braking
device coupled to the simulation. The simulator also incorporates
a four degree-of-freedom non-haptic ultrasound probe simulator.
This next device is a prototype virtual reality interface I
made for training surgeons in performing laproscopic and endoscopic
procedures.

The device below is a prototype virtual reality interface device
I made for training medical personnel in performing needle sticks.

The prototype Kaufman haptic interface for lower urinary tract endoscopic simulation allowed four degrees of freedom and provided a haptic experience to simulate tissue resistance:

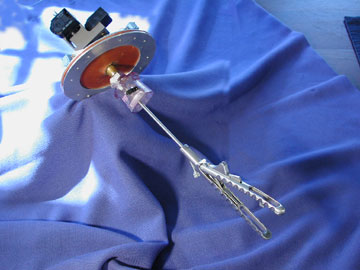
Here are further details of the Kaufman Haptic Catheter Simulator System I designed and built for training interventional radiologists and surgeons in feeding catheters from an incision near the groin either to the brain to treat aneurysms or else to insert filters in the vena cava:
The doctor can push, pull, and twist on the end of the flexible catheter while watching the computer simulation of what they would see on their actual fluoroscopic display. The forces and torques applied by the doctor are transmitted to the computer which displays what would be seen under those conditions in an actual patient. The computer in turn directs the servo system to translate or rotate the catheter as it would behave in an actual patient and to provide the haptic forces and torques that would be felt if the catheter were touching or moving through actual blood vessels or being manipulated to navigate past branches in arteries.
(I designed and built the haptic and electromechanical components seen here along with the device control electronics and computer interfaces but the computer simulation work was done by Robert Lindeman and others within LACAM.)
The above photo shows the actual procedure for inserting and guiding a catheter.
The next photo is a rare historical photo of Professor Kaufman carefully fabricating the catheter simulator using an axe and other stone age tools.


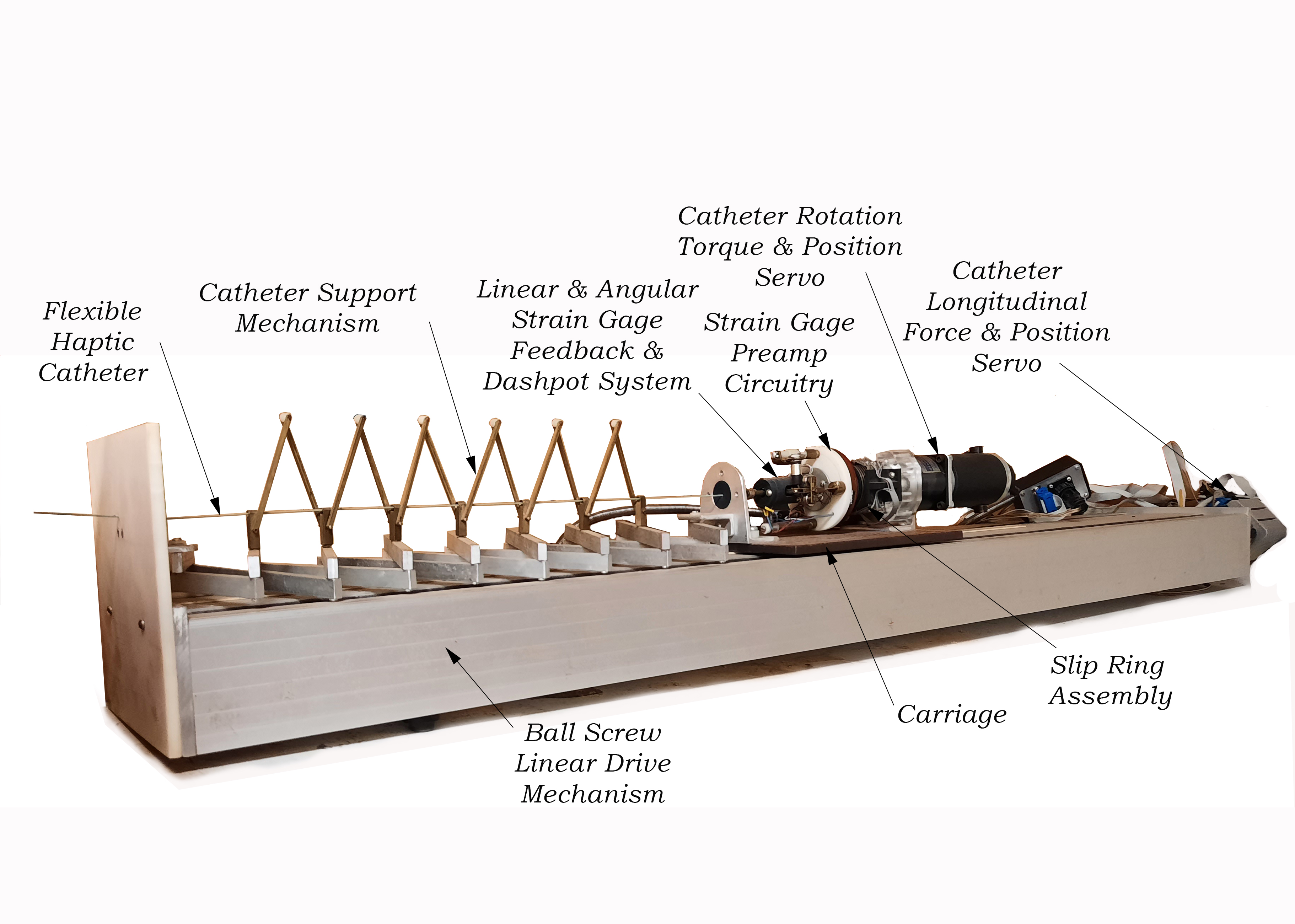
The Haptic Device
The actively controlled electromechanical haptic catheter which serves as the tactile input and output device is unique to this interactive system. Based on the geometry of the patient’s anatomy and the mechanical properties and position of the catheter, the computer can calculate what forces and torques the person holding the catheter should experience. The catheter held by the physician is actively driven so as to produce these tactile sensations. Where the catheter is blocked by collision with a vessel wall the person pushing on the catheter feels an increased resistance. When the catheter is twisted to steer the tip into a branch of the artery, the display shows the new conformation and the user of the system feels the change in position and force. As the interventional radiologist working with the device pushes, pulls, or twists on the end of the catheter, force and torque sensors attached to the catheter measure the stresses being applied. This force/torque sensing module contains a system of strain gages arranged so as to uncouple and orthogonalize the force and torque effects. Forces or torques sensed by the strain gages are converted into electrical signals which are transmitted through slip rings, amplified, and finally converted from analog to digital form. The force and torque sensing module attached to the end of the catheter within the simulated patient rides on an actively controlled torsional drive which controls the angular rotation of the end of the catheter. This twisting mechanism rides, in turn, on a servo motor controlled linear carriage assembly. Shaft position encoders on both the torsional and linear servo motors determine the actual linear and angular positions of the catheter tip and transmit this information to the control computer. The forces and torques applied to the catheter by the radiologist, along with the current linear and angular position of the catheter, are sent to the simulation model. Knowing the vascular geometry and the catheter’s mechanical properties, the simulation sends appropriate data to the visualization and display algorithms and transmits force, torque, and position control commands back to the intermediate control computer. The design of the system allows the control computer to receive one of two kinds of command information back from the modeling computer. It might either be told to correct the forces or torques to some calculated “setpoint” values (as determined by the real-time simulation running concurrently) or else it might be told to correct the linear or angular position of the end of the catheter. In either case, the correction is made by the servo motors pushing, pulling, or twisting on the end of the catheter till the error between the command value and the actual value is driven towards zero. A dynamic model of the physical interaction device resides within the control computer. This model of the “plant” determines the needed corrective action. The feedback loop then drives the linear and rotational servo motors so as to eliminate residual errors in force, torque, or position. By having an active catheter simulation system rather than a purely passive braking device we eventually hope to be able to accurately model fine nuances such as the slight “springback” which a radiologist might experience. This might occur, for example, as the tip of the catheter unsnags when it enters a branched vessel and residual loops or kinks in the catheter suddenly straighten out. We might also be able to model subtle phenomena such as the effect of a catheter touching the pulsating wall of the heart.
Return to Roger
E. Kaufman Homepage












